Environmental Electrochemistry Group lead by Prof. Victoria A. Nikitina specializes on electrochemical energy storage and conversion mechanisms, which will empower the transition from fossil fuel-based economy to renewable energy. We focus at developing metal-ion battery solutions for use under all-climate conditions, electrochemical conversion of carbon dioxide to fuels, green hydrogen production by (photo)electrolysis.
RESEARCH PROJECTS 2022 - 2025
Low temperature ion insertion-based batteries
(Photo)electrochemical hydrogen production
Electrochemical CO2 conversion to fuels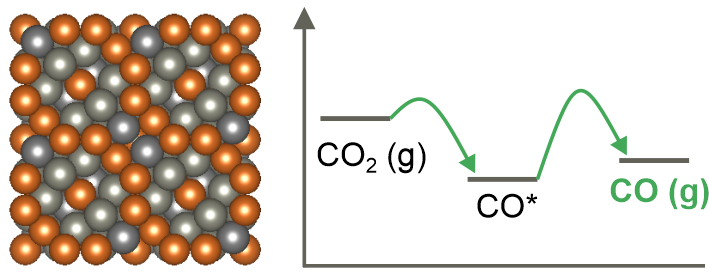
JOIN OUR GROUP
You can join our research group at Skoltech or at the Chemistry Department of Lomonosov Moscow State University. If you are a Bachelor/Master student or a prospective PhD student from Russia or abroad, please email your CV to This email address is being protected from spambots. You need JavaScript enabled to view it. and briefly describe your background and scientific interests.
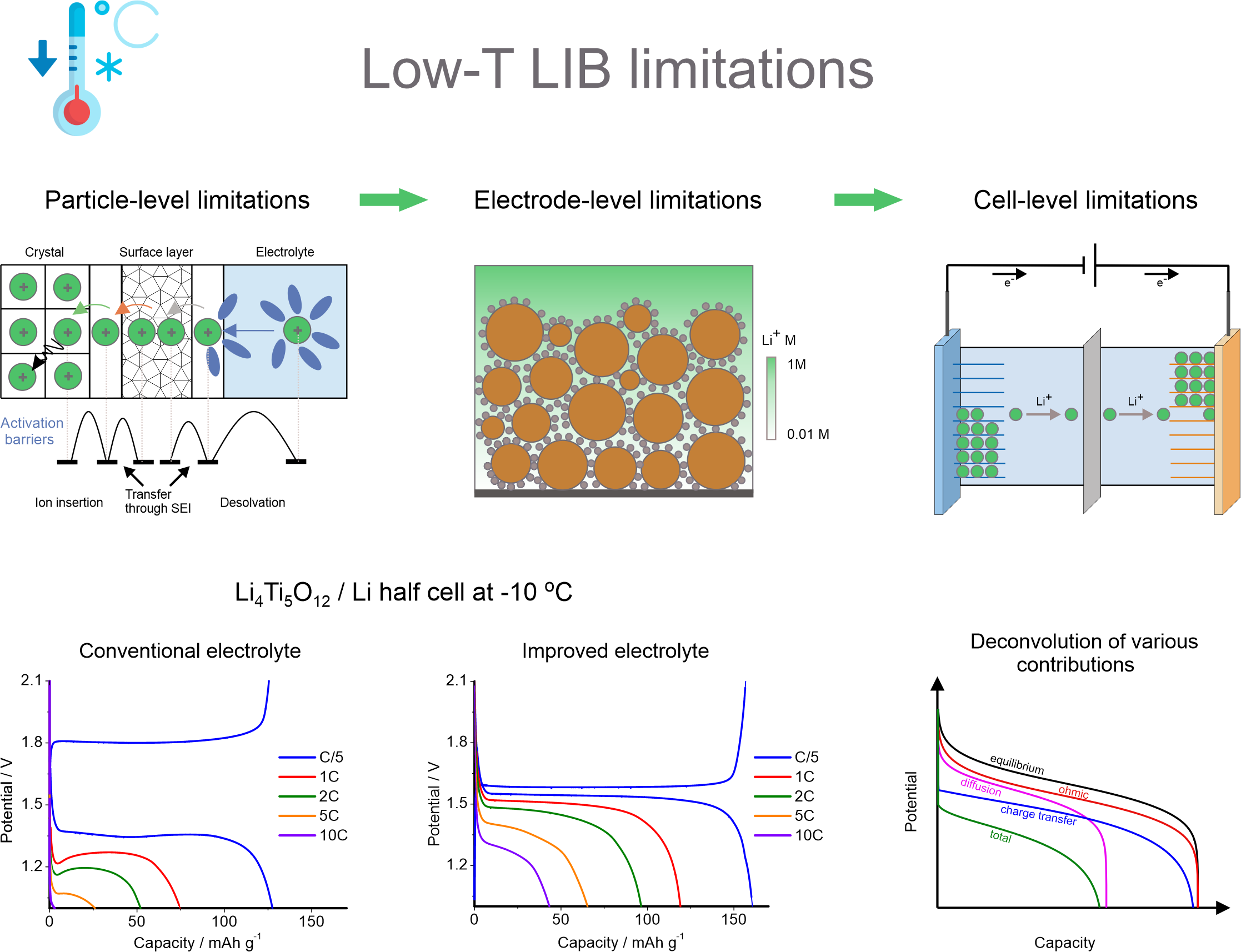
1. Low temperature ion insertion-based batteries
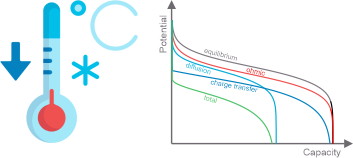
Li-ion batteries degrade quickly at subzero temperatures. To understand the origin of the battery performance deterioration we conduct multifaceted studies of all the factors, which are responsible for the changes in battery kinetics in the extended temperature range: changes in the activation barriers for ion intercalation, changes in the properties of the electrolytes and evolution of the electrode/electrolyte interphases. The ultimate goal is to design temperature-tolerant and safe battery chemistries, which deliver energy in the extended temperature interval (-100 - +100 °C). We focus on both non-aqueous Li-ion and Na-ion chemistries and aqueous rechargeable batteries (proton insertion batteries). Our current projects are:
1. Design of novel low-temperature electrolytes for Li-ion and Na-ion batteries to enable efficient operation in the extended temperature range.
2. Elucidating ion intercalation mechanisms using advanced electrochemistry and spectroscopy to derive information on the relevent rate-limiting steps.
3. Design of aqueous rechargeable proton-based batteries for ultra-high rate charge storage in the extended temperature range.
Recent publications
[1] N.S. Buryak, D.V. Anishchenko, E.E. Levin, S.V. Ryazantsev, V. Martin-Diaconescu, M.V. Zakharkin, V.A. Nikitina, E.V. Antipov, High-voltage structural evolution and its kinetic consequences for the Na4MnV(PO4)3 sodium-ion battery cathode material. J. Power Sources, 518 (2022) 230769. https://doi.org/10.1016/j.jpowsour.2021.230769
[2] A.I. Komayko, S.V. Ryazantsev, I.A. Trussov, N.A. Arkharova, D.E. Presnov, E.E. Levin, V.A. Nikitina, The Misconception of Mg(2+) Insertion into Prussian Blue Analogue Structures from Aqueous Solution. ChemSusChem, 14 (2021) 1574-1585. https://doi.org/10.1002/cssc.202002916
[3] P.M. Chekushkin, I.S. Merenkov, V.S. Smirnov, S.A. Kislenko, V.A. Nikitina, The physical origin of the activation barrier in Li-ion intercalation processes: the overestimated role of desolvation. Electrochim. Acta, 372 (2021) 137843. https://doi.org/10.1016/j.electacta.2021.137843
[4] V.A. Nikitina, S.Y. Vassiliev, K.J. Stevenson, Metal-Ion Coupled Electron Transfer Kinetics in Intercalation-Based Transition Metal Oxides. Adv. Energy Mater., 10 (2020) 1903933. https://doi.org/10.1002/aenm.201903933
[5] V.A. Nikitina, Charge transfer processes in the course of metal-ion electrochemical intercalation. Curr. Opin. Electrochem., 19 (2020) 71-77. https://doi.org/10.1016/j.coelec.2019.10.006
[6] D.V. Anishchenko, M.V. Zakharkin, V.A. Nikitina, K.J. Stevenson, E.V. Antipov, Phase boundary propagation kinetics predominately limit the rate capability of NASICON-type Na3+xMnxV2-x(PO4)3 (0≤x≤1) materials. Electrochim. Acta, 354 (2020) 136761. https://doi.org/10.1016/j.electacta.2020.136761
[7] S.Y. Vassiliev, V.V. Sentyurin, E.E. Levin, V.A. Nikitina, Diagnostics of lithium-ion intercalation rate-determining step: Distinguishing between slow desolvation and slow charge transfer. Electrochim. Acta, 302 (2019) 316-326. https://doi.org/10.1016/j.electacta.2019.02.043
2. (Photo)electrochemical hydrogen production
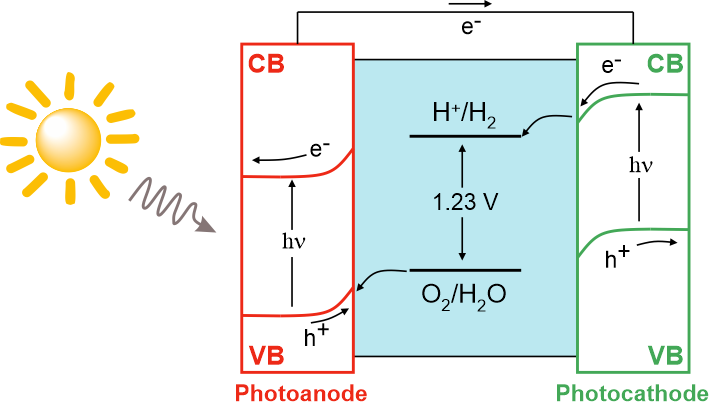
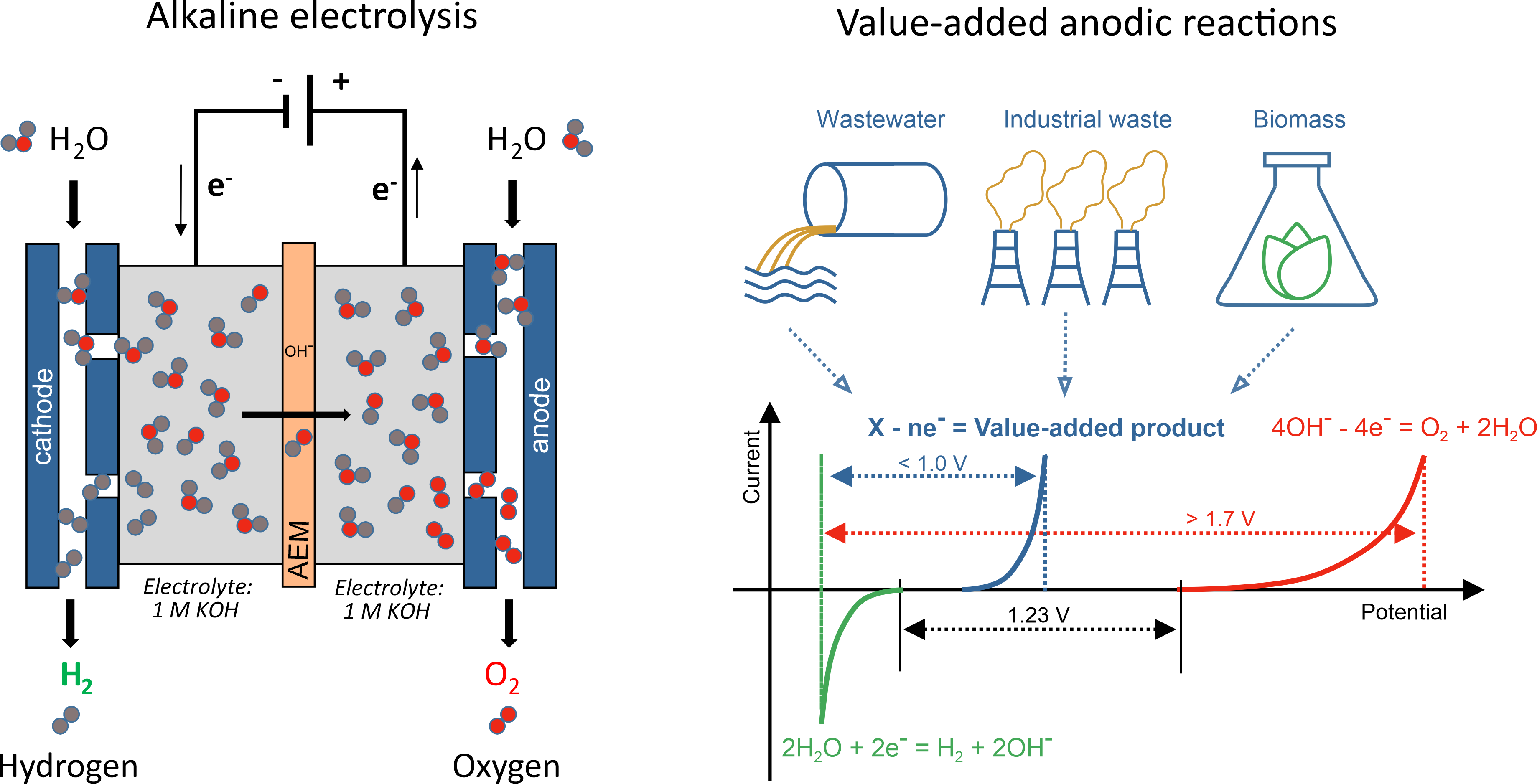
Green hydrogen, produced by electrolysis of aqueous solutions using renewable/sustainable energy sources, is a very much desired energy carrier, which could reduce our dependence on fossil fuels and decarbonize the industry. To date, 96% of hydrogen gas is still obtained from steam reforming and coal gasification, which leads to greenhouse gases emissions. Hydrogen produced by either acidic or alkaline electrolysis is still too expensive. The minimal voltage to break water molecules into hydrogen and oxygen is 1.23 V, while reaching industrially relevant current densities requires much higher voltages (more than 1.7 V). The cost of electricity thus plays a decisive role in the electrochemical hydrogen production. There is much more to be done to increase the chances for success of hydrogen-based economy. New electrocatalysts based on cheap and earth-abundant elements should be developed (currently most active electrocatalysts are based on Pt and Ir metals). Electrolyzer design must be improved to provide the necessary durability and cost-effectiveness. Using pure deionized water for electrolysis is a problem by itself, as it invokes the necessity of a pre-purification step. In our group, we focus on the following strategies to design efficient (photo)electrolyzers.
- Exploring different anodic reactions
When the anodic reaction switches from oxygen evolution (oxidizing water molecules) to oxidizing “softer” chemicals, we simultaneously decrease the operating voltage and gain a route to obtaining value-added chemicals. Instead of producing oxygen in an electrolyzer, one can produce fertilizers, polymer precursors and even fuels. Sulfides, wastewater, biomass can be oxidized in electrolyzers to increase the efficiency of electrochemical hydrogen production.
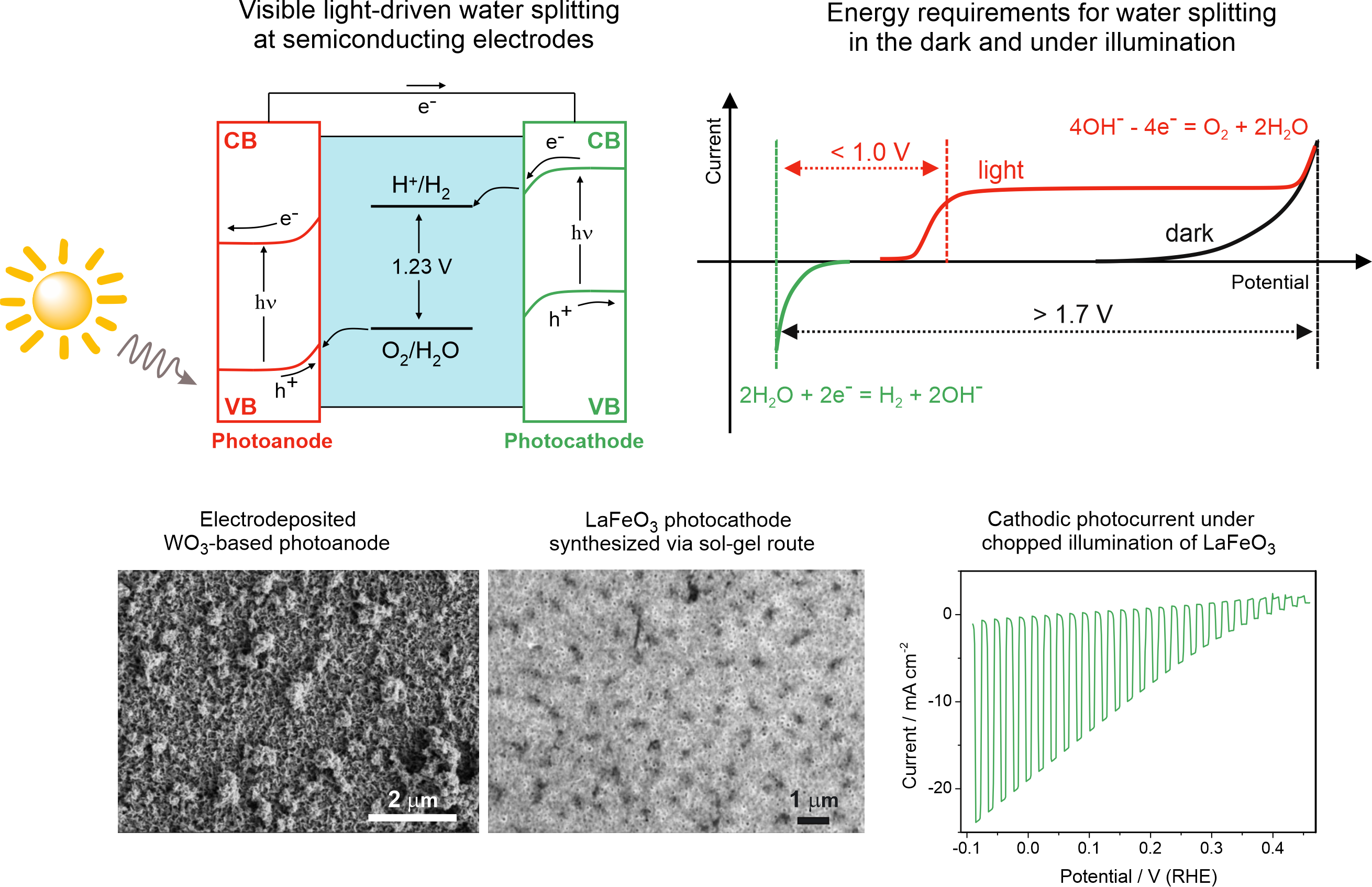
- Photo-assisted hydrogen production
Photoelectrolysis on semiconducting electrodes can ideally eliminate the need for external electricity to conduct electrolysis of aqueous solutions, or at least greatly reduce the voltage required to drive the water splitting reaction. We focus at exploring semiconducting electrodes which directly convert solar energy into chemical bonds. Here, hydrogen is evolving as a cathodic process, while anodic process can be coupled to oxidizing organic species for wastewater treatment or biomass conversion.
Recent publications
[1] S.V. Porokhin, V.A. Nikitina, D.A. Aksyonov, D.S. Filimonov, E.M. Pazhetnov, I.V. Mikheev, A.M. Abakumov, Mixed-Cation Perovskite La0.6Ca0.4Fe0.7Ni0.3O2.9 as a Stable and Efficient Catalyst for the Oxygen Evolution Reaction. ACS Catalysis, 11 (2021) 8338-8348. https://doi.org/10.1021/acscatal.1c00796
[2] S.V. Porokhin, V.A. Nikitina, A.M. Abakumov, Enhancement of Catalytic Activity and Stability of La0.6Ca0.4Fe0.7Ni0.3O2.9 Perovskite with ppm Concentration of Fe in the Electrolyte for the Oxygen Evolution Reaction. Materials, 14 (2021) 6403. https://doi.org/10.3390/ma14216403
[3] V.A. Nikitina, A.A. Kurilovich, A. Bonnefont, A.S. Ryabova, R.R. Nazmutdinov, E.R. Savinova, G.A. Tsirlina, ORR on Simple Manganese Oxides: Molecular-Level Factors Determining Reaction Mechanisms and Electrocatalytic Activity. J. Electrochem. Soc., 165 (2018) J3199-J3208. https://doi.org/10.1149/2.0261815jes
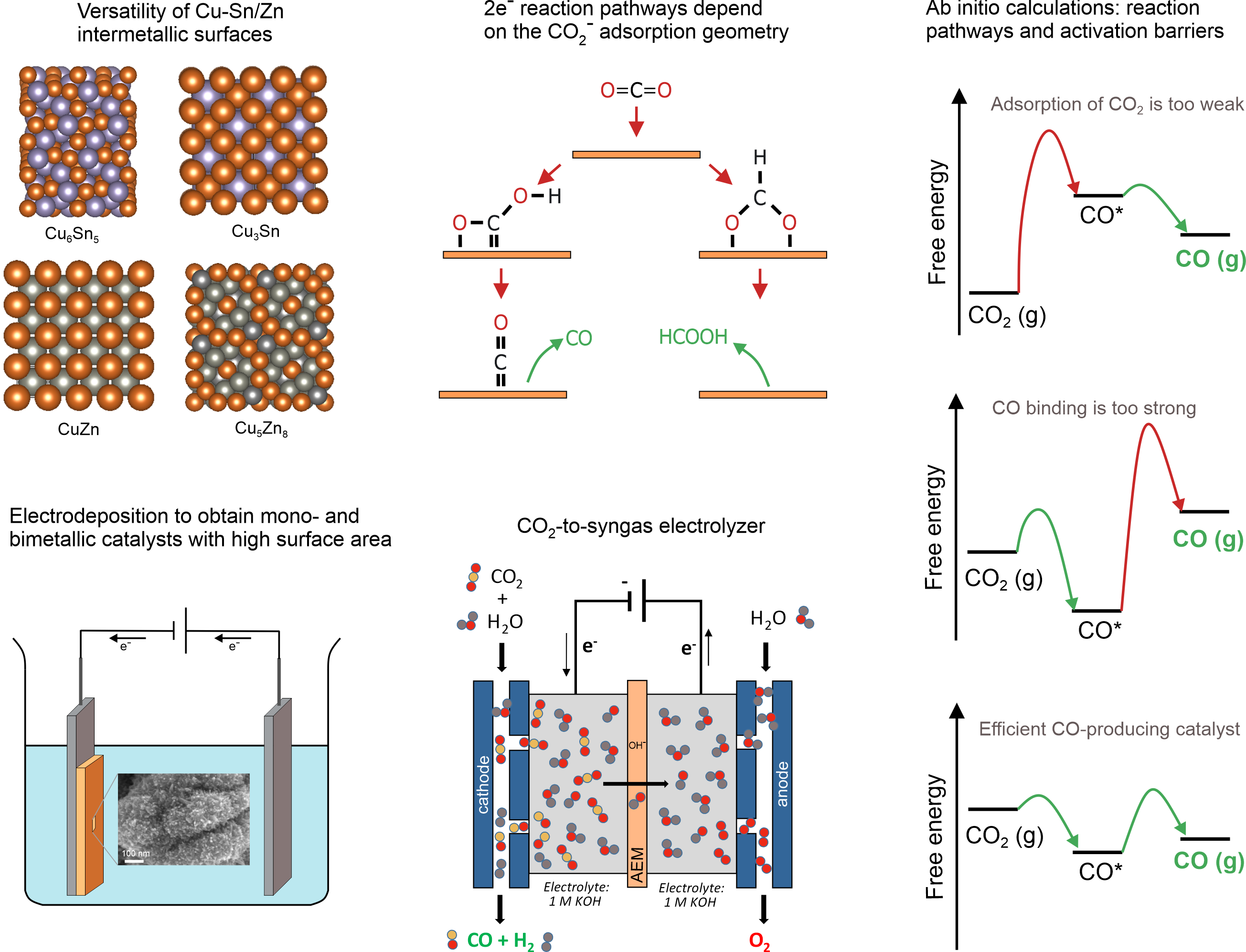
3. Electrochemical CO2-to-fuels conversion
Electrochemical CO2 conversion to fuels and chemical feedstock is an attractive route to reduce carbon dioxide emissions to prevent catastrophic climate changes. We are focused on the design of non-precious metal-based electrocatalysts for CO2 conversion to synthesis gas, formate and alcohols and mechanistic studies of the electrochemical CO2 reduction pathways. We use electrodeposition to create high surface area bimetallic electrocatalysts based on Cu, Sn, Zn to convert greenhouse gas into valuable products.